We are interested in understanding immune-mediated diseases and in strategies to monitor and therapeutically influence the balance between immunity and tolerance. We focus at 3 major topics:
Dendritic cells (DCs) activate T lymphocytes (= T cells), which defend our body against infections with microbes. DCs collect antigens in various tissues, migrate to lymph nodes, and activate the T cells there. T cells then enter various tissues to combat microbes. DCs can also present autoantigens, but this normally leads to immune tolerance by elimination of potentially harmful T cells, or by induction of regulatory T cells that suppress harmful T cells. If such peripheral immune tolerance fails autoimmune diseases result, such as type I diabetes mellitus, multiple sclerosis, rheumatoid arthritis or glomerulonephritis. We apply various techniques like in vivo models for these diseases, cell-specific knockouts, genetic in vivo knockdown, metabolomic analysis, singel cell sequencing and multicolor flow-cytometry and histology to understand the immunopathophysiology.
Here are some of our previous and current research projects:
One of our research foci is the urogenital tract as a target of autoimmunity (Kurts et al, Nat Rev Immunol, 2013). We wish to understand the mechanisms underlying glomerulonephritis and how the defense against kidney infections (pyelonephritis) is regulated. We described DCs in the kidney (Krüger et al, JASN 2004) and demonstrated that they promote glomerulonephritis by presenting glomerular antigen to Th cells (Heymann et al, J Clin Invest 2009; Hochheiser et al, JASN 2011). This explained why tubulointerstitial infiltrates are associated with nephritis progression, and identified kidney DCs as a therapeutic target. In the context of the SFB1192, we found that kidney DCs especially depend on the receptor CX3CR1 (Hochheiser et al, J Clin Invest, 2013), identifying a molecular target of glomerulonephritis therapy.
Kidney DCs also activate the inflammasome in response to crystals that precipitate in diseases like primary or secondary hyperoxaluria, leading to irreversible kidney fibrosis (Ludwig-Portugall, Kidney Int 2016). We showed that inflammasome inhibitors can attenuate these diseases, identifying a new therapeutic principle in crystal nephropathy (funded by the SFBTR57, co-speaker C. Kurts). Finally, we collaborate with Z. Abdullah in studying the mechanism of immune-complex glomerulonephritis during chronic viral infections and with N. Garbi in understanding the immune mechanisms in ANCA vasculitis in the context of the SFB237.
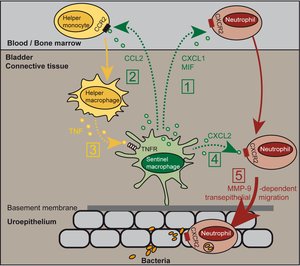
We have generated novel tools to study kidney diseases and DCs (Tittel et al, Nat Methods 2012). We showed that kidney DCs sense invading bacteria in bacterial kidney infection and in response recruit immune effector cells to clear the bacteria (Tittel et al, JASN 2011). We identified a novel immune function of macrophages that use the cytokine TNF to regulate the immune response of neutrophilic granulocytes in bacterial infection (Schiwon et al, Cell, 2014).
Recently, we showed that consuming a high salt diet for only one week weakens the immune system, so that bacterial infections proceed more serious than usual (Jobin et al, Science Transl Med, 2020). This because a high salt diet induces endocrine perturbations that suppress neutrophilic granulocytes, which defend us against bacterial infections.
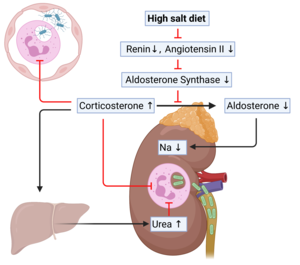
We currently investigate the underlying cell biology in the context of the SFB1454. For a more comprehensive discussion on how salt affects our immune system see our review Jobin et al, Trends Immunol 2021.
The mechanism of cross-presentation enables DCs to activate CD8+ T cells (Kurts et al, Nat Rev Immunol, 2010), which are crucial for the immune defense against viruses, intracellular bacteria and tumors and in vaccination. We identified a new cell biological mechanism of cross-presentation, where antigen is taken up by distinct receptors, into a distinct endosomes presentation (Burgdorf et al, Science 2007), where they are loaded onto MHC I molecules and this process is regulated by TLR/MyD88-signaling (Burgdorf et al, Nat Immunol, 2008).
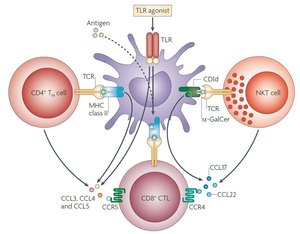
Cross presentation requires that CD4+ Th cells provide a second opinion as to whether an antigen presented by the DC warrants CD8+ T cell activation (= classical cross priming). We have shown that also NKT cells can provide this second opinion (= alternative cross priming). While Th cells induced production of CCR5-binding chemokines, NKT cells caused DCs to produce CCR4-binding chemokines. Both chemokines synergistically enhance cross presentation (Semmling et al, Nat Immunol, 2010). It has been proposed to term these chemokines „Signal 0“, in extension of the classical “Signal 1&2” hypothesis by Bretscher und Cohn. Signal 0 chemokines allow CD8+ T cells to more efficiently locate DCs that present relevant antigen. We currently study whether NKT cells also affect other cellular functions of CD8 T cells, such as their metabolism.
Regulatory T cells (Treg) can control autoreactive T cells. We showed that survival of these cells depends on the NFkB component IKK2. Short-term IKK2 inhibition attenuated glomerulonephritis by preventing DC activation, but long-term inhibition depleted Treg, and aggravated glomerulonephritis (Gotot et al, JASN 2015). This effect can be used to stimulate tumor immunity (Heuser et al, Cell Reports 2017).
We furthermore collaborate with J. Becker-Gotot to study the ability of Treg to prevent autoantibody formation cells via PDL-1 that deletes autoreactive B cells. (Gotot et al, PNAS 2012).
Post-operative ileus (POI) is the most prevalent complication after intestinal surgery and results in serious medical complications and large costs to the health system. We discovered that POI is an immune-mediated disease: gut-resident Th1 memory cells recirculate through the body after surgery and settle in all gut segments. There, they produce IFNg that stimulates resident macrophages to produce nitric oxide, which directly paralyses intestinal muscle cells, causing ileus (Engel et al, Nat Medicine 2010). This mechanism is induced by distinct CD11b+ CD103+ DCs that sense dislocated intestinal microbiome (Pohl et al, Gut 2017).
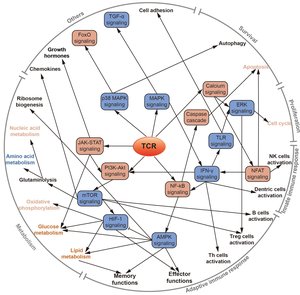
We are studying metabolic consequences of T cell receptor signaling. We have developed novel bioinformatic approaches that allow deciphering the regulation of pathogenic T cell responses in various tissues and disease situations.
Tel.: +49 228 287 - 11050
Fax: +49 228 287 - 11052
ckurts(at)uni-bonn.de
